
Nov 21, 2018
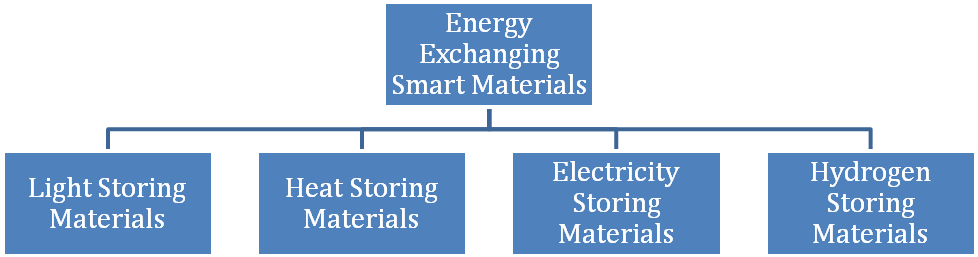
Materials that are able to store latent and sensible energy in the form of light, heat, electricity or hydrogen and exhibit reversibility are known as energy-exchanging smart materials. Thermal energy is stored as a change in internal energy of a material as latent heat, thermochemical, sensible heat or a combination of these.
In sensible heat storage, thermal energy is stocked by raising the temperature of a liquid or solid. Latent heat storage is based on the heat absorption and heat release when a material experiences a phase change from liquid to gas, solid to liquid or vice versa. Phase change can be in the following forms: solid-solid, solid-liquid, solid-gas, liquid-gas and vice versa. In several transitions, such as solid-solid, heat is stockpiled as the material is altered from one crystalline to another. These transitions generally have smaller latent heat and smaller volume changes than solid-liquid transitions. Solid-solid PCMs offer the advantages of less stringent container requirements and greater design flexibility.
Figure 1
Classification of Phase Change Materials
Source: BCC Research
Light and heat storing materials are already commercialized, while electricity and hydrogen storing materials are under development. A number of systems, such as lead accumulators that store electricity, are in use today. In cold conditions, these lead accumulators with reduced capacity exhibit a certain degree of reversibility.
However, as they exhibit reversibility at low temperature and not at normal temperature, these products cannot be used for practical applications. Metal hydrates are most suitable for storing hydrogen, but they lack reversibility and thus cannot be classified as smart materials. However, research is underway on smart hydrogen-storing materials for commercialization.
There are three types of thermal energy storage processes: sensible heat storage (SHS), latent heat storage (LHS) and thermochemical storage. LHS materials that are used to store thermal energy through state changes are known as PCMs. A PCM has the ability to store and release a large amount of heat/energy while maintaining a constant temperature.
Unlike other insulation materials, a PCM, upon reaching its melting point (the temperature at which it changes phase, from solid to liquid), absorbs a large amount of heat and releases its stored latent heat with solidification. PCMs have tremendous potential to fulfill the growing need for energy for cooling and heating applications across various industries, such as construction, commercial refrigeration, textiles and packaging.
Types of Phase Change Materials (PCMs)
PCMs can be broadly classified as organic PCMs or inorganic PCMs.
Organic PCMs are comprised mostly of paraffin, fatty acids and bio-based PCMs. They are not corrosive and are chemically and thermally stable. These materials also have certain drawbacks; they have low melting density and are highly flammable. Bio-based PCMs are obtained from bio-based products. BioPCMs are obtained from a mixture of soy-based chemicals, changes from liquid to solid state and vice versa at a given temperature. These products are used in residential and commercial structures, helping these structures maintain a certain temperature.
Inorganic PCMs contain salt hydrates. Unlike organic PCMs, these materials have high melting enthalpy and high density. With their unique structural combination, these materials are highly corrosive and have relatively less stability.
Paraffin wax (organic PCM) and salt hydrates (inorganic PCM) are the most commonly used PCM materials. Inorganic PCMs are widely used because they have higher melting enthalpy and sharper melting points than organic and eutectic PCMs.

Biophotonics: Technologies and Global Markets (PHO024B)

Global Trade: A Strategic Shift The global trade environment is undergoing a dra...

The global demand for cutting-edge materials continues to rise, and at the foref...

We are your trusted research partner, providing actionable insights and custom consulting across life sciences, advanced materials, and technology. Allow BCC Research to nurture your smartest business decisions today, tomorrow, and beyond.
Contact UsBCC Research provides objective, unbiased measurement and assessment of market opportunities with detailed market research reports. Our experienced industry analysts assess growth opportunities, market sizing, technologies, applications, supply chains and companies with the singular goal of helping you make informed business decisions, free of noise and hype.