
Oct 26, 2017
Blog Market Research Upcoming Event: Translational Sciences Point to Major Market Opportunities
Anyone invested in the growing sector of personalized, or "precision" medicine, pharmaceuticals, medical devices and diagnostics will want to attend “Companion Diagnostics & Personalized Medicine,” a market research seminar, networking and Q&A event in San Diego, California on November 9.
Below you’ll find a few discussion highlights from Senior Editor Robert G. Hunter. BCC Research Members can attend for free. Additionally, limited comp tickets are still available to parties considering BCC Research membership.
These innovations are fueling a pipeline from research to clinical applications.
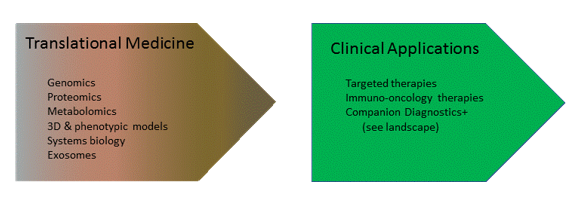
Technology enablers include sequencing (NGS, (dd)PCR) and liquid biopsy, which encompasses many different approaches (cfDNA, exosome, miRNA), resulting in hyper segmentation and requiring strategists to maintain knowledge across several translational sciences/markets as well as a dynamic companion diagnostics (CDx) landscape, which is growing > 30% CAGR.
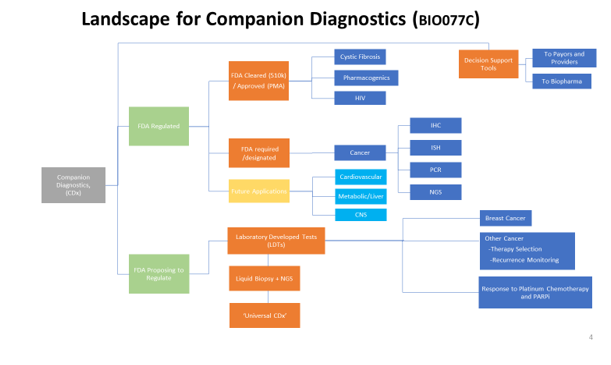
In some cases, translational disciplines are opening new research breakthroughs. For example, exosomes and others are fueling liquid biopsy growth at a > 100% CAGR. This example will be discussed in detail, covering technologies, applications, markets, trends and implications by Dr. Marianna Tcherpakov (see bio).
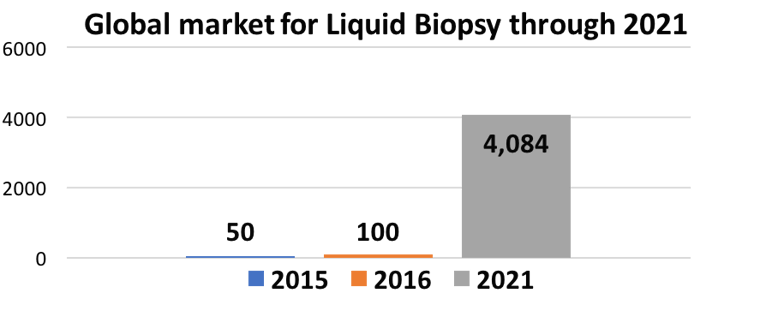
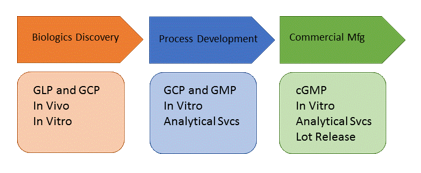
 New horizons: Biologics
New horizons: Biologics
As Immuno-oncology grows rapidly in the near-future, to support development and manufacturing of these biologics, we predict that testing of biologics will be a major focus of innovation, to mature and scale the commercial landscape.
These insights come from our forthcoming report on Biologics Safety Testing (Jan ’18), and an overview will be presented.
About the Speakers:
Dr. Marianna Tcherpakov, Market Analyst
Dr. Marianna Tcherpakov has more than 10 years of experience as a bench scientist specializing in the areas of biochemistry, cell biology and industrial assay development and drug manufacturing. She has contributed to a number of scientific publications and holds several patents. She has expertise as an assay development scientist and is familiar with different research field trends and likely future developments.
In the last five years, Marianna worked in business development sector for variety of biotech companies. She has extensive knowledge in marketing and client management in different segments of pharma and biotech industry including antibody production, gene editing and GMP manufacturing.
Robert G. Hunter, Senior Editor- Healthcare
Robert G. Hunter is a senior editor, technology and market analyst and management consultant with over 20 years in life sciences and healthcare, including formerly Senior Manager in Deloitte Consulting’s Life Sciences Practice. With recent focus on drug discovery & development technologies, Bob has conducted extensive research, analysis, and interviews enabling insights on the emerging research landscape as well as forward-thinking strategies for expanding to clinical applications such as precision cancer therapies, new CNS approaches and regenerative medicine. Sample of projects:
*Market analysis, commercialization consulting and forecasting of customer adoption/revenue for high content screening (HCS) leader
*Discovery IT Roadmap for vaccines (biologics) unit of mid-sized pharma
*Market/technology research and business development for $7B metabolomics market, $5 B in vitro and in silico market, and $400 M 3D cell culture market. Developed thought-leadership and maintain industry forecasts and business relationships.
*3D cell culture technology/market assessment for precision cancer therapy
*Stem cell lines & imaging assay for true genetic diversity
*Proteomics-based biomarkers using label free imaging
*Product development and commercialization plan for Worcester Polytechnic incubator for in vitro diagnostic and tissue applications. Previously, served as interim CEO of spinout.
*Market analysis, commercialization and product development planning for spinout of Clemson University in medical simulation and clinical training applications. Previously, helped to vet various technologies e.g. biomaterials, 3D cell culture, tissue engineering, and regenerative medicine.

Amending the Pesticide Trade Business China and the U.S. are the foremost export...

Introduction The electric vehicle (EV) revolution is not solely about batteries ...

In the ever-evolving landscape of technology, pressure sensors have emerged as p...

We are your trusted research partner, providing actionable insights and custom consulting across life sciences, advanced materials, and technology. Allow BCC Research to nurture your smartest business decisions today, tomorrow, and beyond.
Contact UsBCC Research provides objective, unbiased measurement and assessment of market opportunities with detailed market research reports. Our experienced industry analysts assess growth opportunities, market sizing, technologies, applications, supply chains and companies with the singular goal of helping you make informed business decisions, free of noise and hype.